Chimica organica/RNA: differenze tra le versioni
| Riga 87: | Riga 87: | ||
[[Image:PhosphodiesterBondDiagram.png|thumb|200px|Diagram of phosphodiester bonds between nucleotides]] |
[[Image:PhosphodiesterBondDiagram.png|thumb|200px|Diagram of phosphodiester bonds between nucleotides]] |
||
Dal punto di vista chimico, il DNA e l'RNA sono le forme polimeriche dei nucleotidi. Il legame che unisce i nucleotidi è il '''legame fosfodiestere''', un legame di tipo estereo tra il fosforo del gruppo fosfato di un nucleotide con il carbonio 3' dello zucchero di un altro nucleotide. |
|||
A '''phosphodiester bond''' is a group of strong covalent bonds between the phosphorus atom in a phosphate group and two other molecules over two ester bonds. Phosphodiester bonds are central to all life on Earth, as they make up the backbone of the strands of DNA. In DNA and [[RNA]], the phosphodiester bond is the linkage between the 3' carbon atom and the 5' Carbon of the sugar ribose. |
|||
I gruppi fosfato che danno vita al legame fosfodiestere sono caratterizzati, a pH 7, da una intensa carica negativa. Questo è dovuto al fatto che la pK<sub>a</sub> dei gruppi fosfato è prossima allo zero. La repulsione dei gruppi fosfato causata da tale carica fa sì che tali gruppi si dispongano alle estremità opposte del filamento del DNA. La carica è neutralizzata da proteine (gli istoni), ioni metallici e poliamine. |
|||
The phosphate groups in the phosphodiester bond are very negatively-charged. Because the phosphate groups have a pK<sub>a</sub> near 0, they are negatively-charged at pH 7. This repulsion forces the phosphates to take opposite sides of the DNA strands and is neutralized by proteins (histones), metal ions, and polyamines. |
|||
In order for the phosphodiester bond to be formed and the nucleotides to be joined, the tri-phosphate or di-phosphate forms of the nucleotide building blocks are broken apart to give off energy required to drive the enzyme-catalyzed reaction. When a single phosphate or two phosphates known as pyrophosphates break away and catalyze the reaction, the phosphodiester bond is formed. |
|||
Hydrolysis of Phosphodiester bonds can be catalyzed by the action of phosphodiesterases which play an important role in repairing DNA sequences. |
|||
In biological systems, the phosphodiester bond between two ribonucleotides can be broken by alkaline hydrolysis because of the free 2' hydroxyl group. |
|||
[[Categoria:Chimica organica|RNA]] |
[[Categoria:Chimica organica|RNA]] |
||
Versione delle 00:16, 20 ago 2008
Per descrivere l'acido desossiribonucleico (DNA) e l'acido ribonucleico (RNA) dal punto di vista chimico è opportuno partire dagli elementi di base che lo costituiscono, le basi azotate.
Le basi azotate
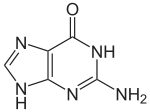
Con il termine di basi azotate in biochimica ci si riferisce alle basi puriniche Adenina e Guanina e alle basi pirimidiniche Citosina, Timina (presente solo nel DNA) e Uracile (presente solo nell'RNA).
| Basi azotate puriniche | Basi azotate pirimidiniche | |||
|---|---|---|---|---|

|
|

|

|

|
| Adenina | Guanina | Citosina | Timina | Uracile |
I nucleosidi
Con il termine di nucleoside si indica una glicosilamina formata da una base azotata e un pentoso, D-ribosio (nell'RNA) o 2-desossi-D-ribosio (nel DNA). La base e lo zucchero sono legati con legame N-glicosidico, cioè un legame glicosidico in cui il nucleofilo è rappresentato da un azoto.
I nucleotidi

I nucleotidi sono gli esteri fosforici dei nucleosidi e sono costituiti dunque da un gruppo fosfato, da una base azotata e da ribosio o deossiribosio. La presenza del residuo fosforico conferisce carattere fortemente acido ai nucleotidi ( per questo noti anche come acido adenilico o acido guanilico )
I nomi dei nucleotidi sono abbreviati in codici standard di tre o quattro lettere.
- Una d iniziale sta per "desossi-", cioè indica che il nucleotide in questione è un desossiribonucleotide. Nel caso di un ribonucleotide la d non compare.
- La seconda lettera indica il nucleoside corrispondente alla nucleobase:
- G sta per Guanina
- A sta per Adenina
- T sta per Timina
- C sta per Citosina
- U sta per Uracile
- La terza e la quarta lettera indicano la lunghezza della catena di gruppi fosfato attaccata (mono-, di- e tri-, abbreviati in M, D, T) e la presenza del gruppo fosfato (P).
Ad esempio, la desossicitidina trifosfato è abbreviata con dCTP (la d perché è un desossiribonucleotide, la C perché è una citidina, TP perché è un tri-fosfato).
I nucleotidi sono i monomeri degli acidi nucleici (DNA e RNA).
DNA e RNA
| Queste sono solo alcune immagini, che presto verranno corredate da testo | ||
|---|---|---|
|
 |

|

|
File:AUHoogsteen.png | |
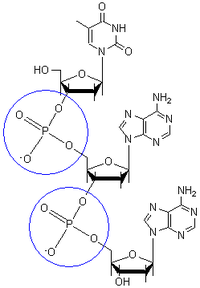
Dal punto di vista chimico, il DNA e l'RNA sono le forme polimeriche dei nucleotidi. Il legame che unisce i nucleotidi è il legame fosfodiestere, un legame di tipo estereo tra il fosforo del gruppo fosfato di un nucleotide con il carbonio 3' dello zucchero di un altro nucleotide. I gruppi fosfato che danno vita al legame fosfodiestere sono caratterizzati, a pH 7, da una intensa carica negativa. Questo è dovuto al fatto che la pKa dei gruppi fosfato è prossima allo zero. La repulsione dei gruppi fosfato causata da tale carica fa sì che tali gruppi si dispongano alle estremità opposte del filamento del DNA. La carica è neutralizzata da proteine (gli istoni), ioni metallici e poliamine.


